Examining proteins, nucleic acids and other biomolecules is not a simple task. Therefore scientists often use photochemically controlled bioorthogonal reactions for their labelling. Tomáš Slanina, an Experientia Foundation grant recipient, devoted his research stay at Goethe University in Germany to the study of this very issue. Today, intensive research is under way looking into possible uses of non-coding RNA as a pharmaceutical. This is a crucial field of research as non-coding RNA could be applied in the treatment of a wide range of diseases. “There are studies of the importance of microRNA as a key to angiogenesis or in cell proliferation or as a part of cancer research. We use biological mice infarct models in which we try to promote tissue regeneration after a heart attack using microRNA-34a,” says Tomáš Slanina.
You study photochemically controlled bioorthogonal reactions in your research. Can you explain what reactions are they?
Bioorthogonal reactions do not happen inside a test tube, they occur in biological conditions – in water, inside live cells, in the presence of other biomolecules and other functional groups. Our bodies too consist of more or less reactive biomolecules. Bioorthogonal reactions have been developed so that we could control biomolecules in their natural environment. Most reactions that we know are thermal reactions. But we study reactions that work on photochemical principles. Such reaction mixtures need to be irradiated. The reaction is powered by the energy of the absorbed photons. At the moment you stop the irradiation, photochemical process ends. It means that we can control exactly when and where the reaction will start. We can aim the laser at a certain area in an organism to trigger the required change.
You mentioned that you are trying to prepare products that can be activated by visible light. What is the principle of your project?
We work with coloured substances – this is necessary if we want to activate them by visible light. We use well-known dyes that absorb light very well and are modified so that they subsequently break down in a pre-defined manner. A photo-activatable agent (so-called linker, Fig. 1) is designed to have functional groups at both ends that can trigger bioorthogonal reactions with a modified biomolecule, in particular with an oligonucleotide (modified RNA) whose sequence of bases is complementary to that of a particular microRNA. A modified RNA is prepared using basic building blocks by a solid-phase synthesis. It is substituted with an alkyne (or its precursor) at both ends and can therefore react with a photo-activatable linker that can cyclize it under optimized reaction conditions (Fig. 1). The cyclic RNA is biologically inactive and can be applied into an organism without triggering any changes of biochemical processes. It does not form complexes with other nucleic acids mediated by Watson-Crick interactions.

Figure 1: A modified RNA is prepared by a solid-phase synthesis from building blocks (left). The modified biomacromolecule then reacts in a bioorthogonal reaction with a photo-activatable linker that cyclizes it. The cyclic RNA does not interact with the complementary microRNA in the organism (right).
MicroRNAs are very important regulators in an organism. By adding a complementary chain to the target microRNA we can switch off its activity. RNA is inherently single-stranded. When we add a complementary twin, they hybridise, resulting in the occurrence of a dimer. Such RNA duplex is inactive and does not take part in biochemical processes (Fig. 2). In order to control this regulation by light, we use photo-activatable cyclic RNAs that are applied directly into cells and tissues. At the moment a part of the tissue is irradiated with a red laser, the linker breaks down at that place (Fig 2) and linear RNA is created. The photoproduct of the linker break-down is fluorescent which is convenient for monitoring of the released linear RNA using fluorescent display techniques. This method not only enables spatial and temporal control over on-off switching of the microRNA function, but it also makes it possible to watch it live.
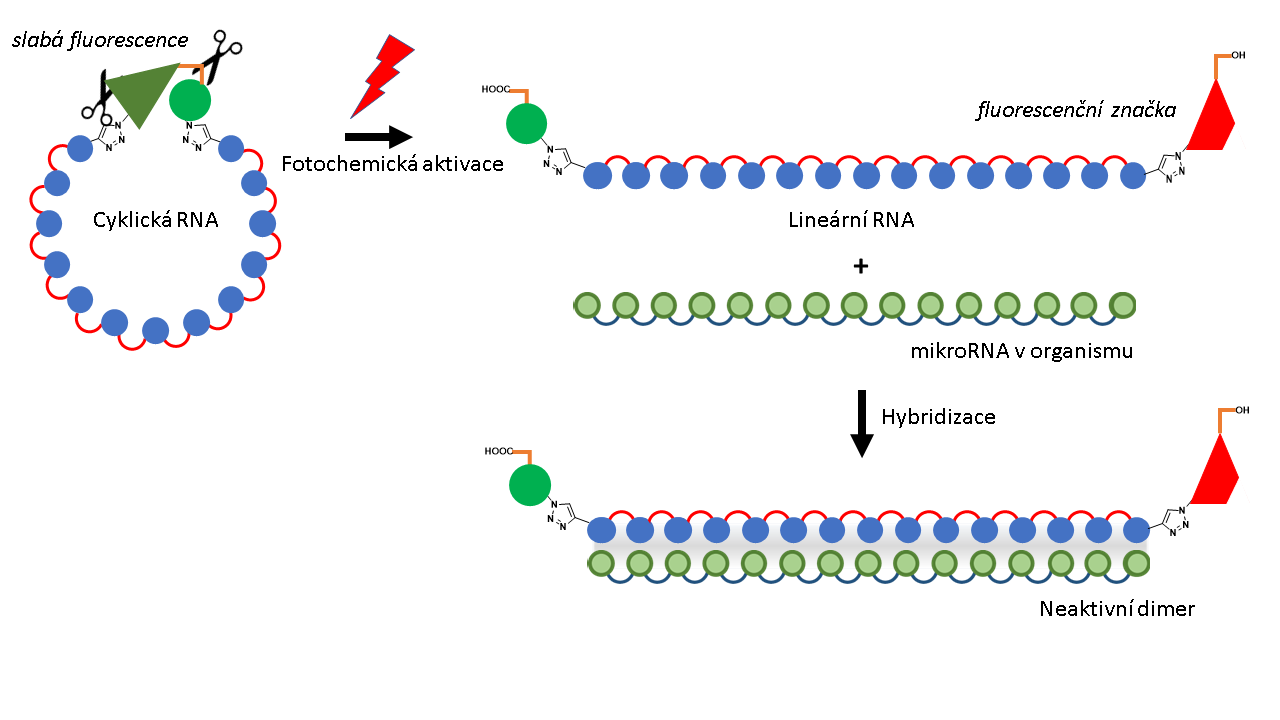
Figure 2: Visible light (red lightning symbol) splits the slightly fluorescent linker in two places (scissors symbol). This results in a linear RNA labelled with a fluorescent label (red trapezoid). The linear RNA is conformationally flexible and can hybridise with microRNA in an organism and block its function by producing an inactive dimer (bottom right).
Both biological and chemical applications of bioorthogonal reactions are very interesting. Where can they be used?
They are mostly used in basic scientific research. The study of biomolecules alone is very complicated, and they need to be labelled somehow in order for us to be able to study the way they distribute in a cell or an organism, how fast different transformations happen etc. Labelling is often done using labels that need to be somehow incorporated in these molecules. Bioorthogonal reactions turned out to be one of the most suitable method. The process consists of preparing a molecule with a bioorthogonally reactive functional group and subsequently attaching a label, such as a fluorescent dye, to that particular place. A protein or a nucleic acid labelled in this way can be studied, for example by using fluorescence microscopy.
What can your discovery bring to the pharmaceutical industry?
The use of non-coding RNA as a pharmaceutical is now the subject of a very intensive research. One of the possible applications is a simple administration of RNA. By either increasing or blocking the level of microRNA using a complementary anti-miRNA strand, we can regulate a number of signalling pathways and functions in an organism. This is a biogenic method of making the organism do something what it is not programmed to do at the given moment. By directly interfering in regulatory mechanisms, we are creating a therapeutic with a completely different mechanism of action than a vast majority of medications used so far. It can fine-tune the concentration of certain proteins and deliver them at desired destinations.
What diseases could be treated with the help of this therapeutic?
Our approach is very general and therefore it can aim at a wide range of diseases. There are for instance studies looking into the importance of microRNA in angiogenesis, cell proliferation, or cancer research, but it can be also used in case of inflammatory diseases. We use biological mice infarct models in which we try to promote tissue regeneration after a heart attack using microRNA-34a regulation. MicroRNA-34a is a rather dangerous strand, therefore its concentrations must be properly regulated. We are trying to regulate its concentration in tissue to produce the desired effect without any side effects.
Does it make your research multidisciplinary then?
Certainly. We start with theoretical chemistry – the molecule design is a rational design based on photo-physical properties that the target molecule should have. We then continue with synthetic organic chemistry, followed by physicochemical measurements of the molecule parameters (what light they absorb, how efficiently is their fluorescence produced, in what manner, how efficiently and what products they break down into etc.). Then comes the chemico-biological part of the research where my colleagues and I synthetize biomacromolecules, oligonucleotides in particular, which are subsequently functionalized and cyclized. Finally, we cooperate with the University Hospital in Frankfurt where they carry out tests on biological models for activation in blood, metabolism, biostability, toxicity etc.
Why have you specialised in this area of chemistry?
I have been fond of organic chemistry since my first year in the bachelor’s programme at Masaryk University in Brno, I found it the most interesting field of science. In organic chemistry you need not to have an encyclopaedic knowledge to understand the basic principles. I would compare it to mathematics. And when you master certain laboratory skills, you are ready to synthetize, clean and analyse your molecules. I have always liked that I can do chemistry directly, not just on a computer screen. At length I specialised in photochemistry, i.e. the activation of molecules using light. I started in the research group of Professor Klán, a photochemist himself, and I grew very fond of this field and I have held onto photochemistry ever since. During my research stays I have studied those areas of chemical biology where photochemistry is employed in a different way, but it is also very important. I can see a great future in biosciences in general, therefore I strongly believe in interdisciplinarity.
You have been awarded the Experientia Foundation grant and could spend one year in the research group of Professor Heckel at Goethe University in Germany. Why did you find Professor Heckel’s research appealing?
He has appealed to me because he combines photochemistry with a field of chemical biology which was completely new for me. Working with biomolecules seemed very exotic to me and I was telling myself I would really love to learn something like this. I had no desire to do research in a lab where they specialise only in chemical biology because I wanted to contribute my own knowledge to the group as well. And this happened with Professor Heckel. We could both profit from what we know and the result was a really beautiful project. This made it possible for me to stay with his group a half year longer.
What does the research team of Professor Heckel work on and what part of his research were you in charge of?
The team looks into several research topics, all of them studying the use of light in the regulation of biomolecule function. There are different ways of regulation – one is RNA cyclization which is our task, but we also have fellow doctoral students who study the regulation of ion channels in membranes using light, or DNA nanoarchitecture, ligand synthesis in the regulation of riboswitch-based RNA expression, or they develop new photo-activatable molecules.
What was it like to work with Professor Heckel? What system does he use in his work and what crucial things have you learned from him?
Professor Heckel is a young, dynamic professor and his enthusiasm for science is contagious. He gave me absolute liberty in what I wanted to do, and, on the other hand, when I needed advice he was always around. He helped me meet plenty of people who are experts in the field and whom I could consult. He inspired me to that extent that I started seriously thinking of the academic career.
And what were your impressions of Goethe University in Germany? You already had some experience from the joint PhD programme in Regensburg. How is the German scientific community different from the Czech one?
It is very different. In Germany, 95% of chemistry graduates continue their studies in a PhD programme, on a mass scale I would say. Also, 95% of German PhD graduates look for work in an industrial sector. Having completed their PhD they never want to set foot in a lab again. Most of them are looking for managerial positions in the chemical industry. Their motivation is completely different from what we do. We followed our hearts, we were science enthusiasts, here it is just one more thing to tick off on your to-do list in life.
What were your goals when you went on your research stay?
I went on my research stay with a goal to persuade the science to work. We were really lucky that we have accomplished it which makes me happy.
You have had a publication in the Journal of the American Chemical Society during your stay. What is it about?
The publication called “In Search of the Perfect Photocage: Structure–Reactivity Relationships in meso-Methyl BODIPY Photoremovable Protecting Groups” looks into the development of new photoremovable protecting groups on the basis of BODIPY (fluorescent dye, ed. note) and I think its impact will be huge. We have succeeded in optimising a group of molecules to allow for a nearly hundred percent quantum efficiency of the chemical reaction occurring after the visible light absorption. This is absolutely exceptional; nothing of the kind exists, at least as far as we know.
Many scholars have prolonged their stay abroad, you among them. What are your plans for the future?
I am heading for another post-doctoral research stay in the group of Professor Ottosson in Uppsala, Sweden. He also engages in the study of photochemistry but from a different perspective. He studies excited state aromacity of molecules.
Can you tell us in conclusion what the Experientia Foundation grant has meant for you personally and what progress you have made in your career, and also as a person, thanks to the research stay?
For me it was an opportunity to work for 12 months in a research project that I designed and lead myself and nothing and nobody limited me. I felt very committed to the Experientia Foundation because of this absolute liberty. I wanted to prove that I take science seriously and I wanted to achieve the best results possible. It definitely is a catalyst in your career. It also changed the way I think about the world of science and my future.
What message would you like to give to prospective applicants for the Experientia Foundation grant?
I keep my fingers crossed for them; I would recommend it to everybody. I have to say that both the application as such and the paperwork process really are a piece of cake. I would also recommend to all future grant recipients when they are negotiating the conditions of their position that they should be asking from the very beginning about the possibilities of internal funding at the hosting university, because one year is a very short period of time if you take it seriously.

Tomáš Slanina
born in Brno in 1988. He earned his master’s degree in organic chemistry at Masaryk University in Brno. He completed a joint PhD programme at the above mentioned MU in Brno and the University of Regensburg in Germany. Thanks to the grant in the amount of CZK 870,000 awarded by the Experientia Foundation he could pursue his studies abroad at Goethe University in Frankfurt, Germany, in a post-doctoral research stay in the group of Professor Heckel. The project was successful and Professor Heckel offered him to stay in Germany for another half-year. At present, Tomáš is heading to Uppsala, Sweden, to his next post-doctoral research stay.